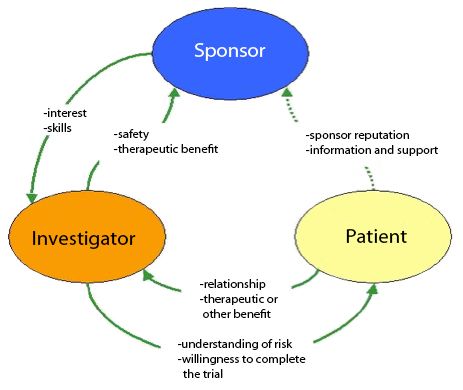
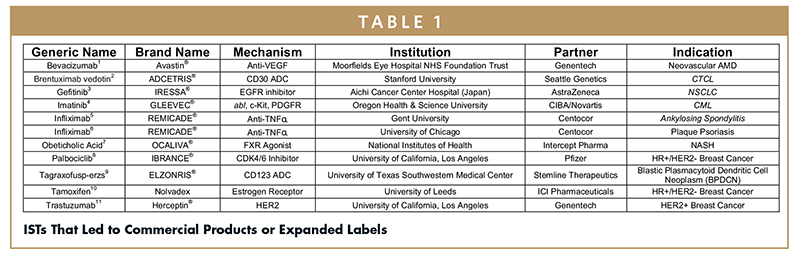
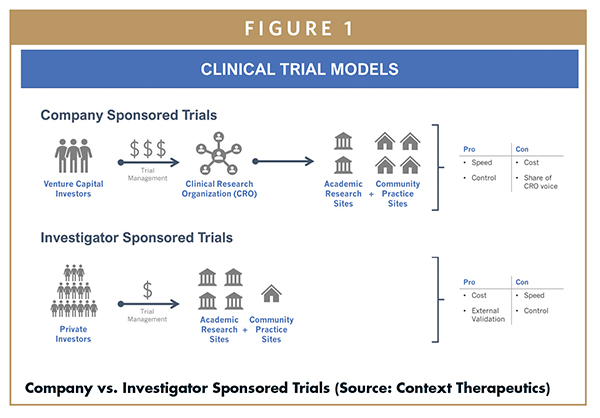
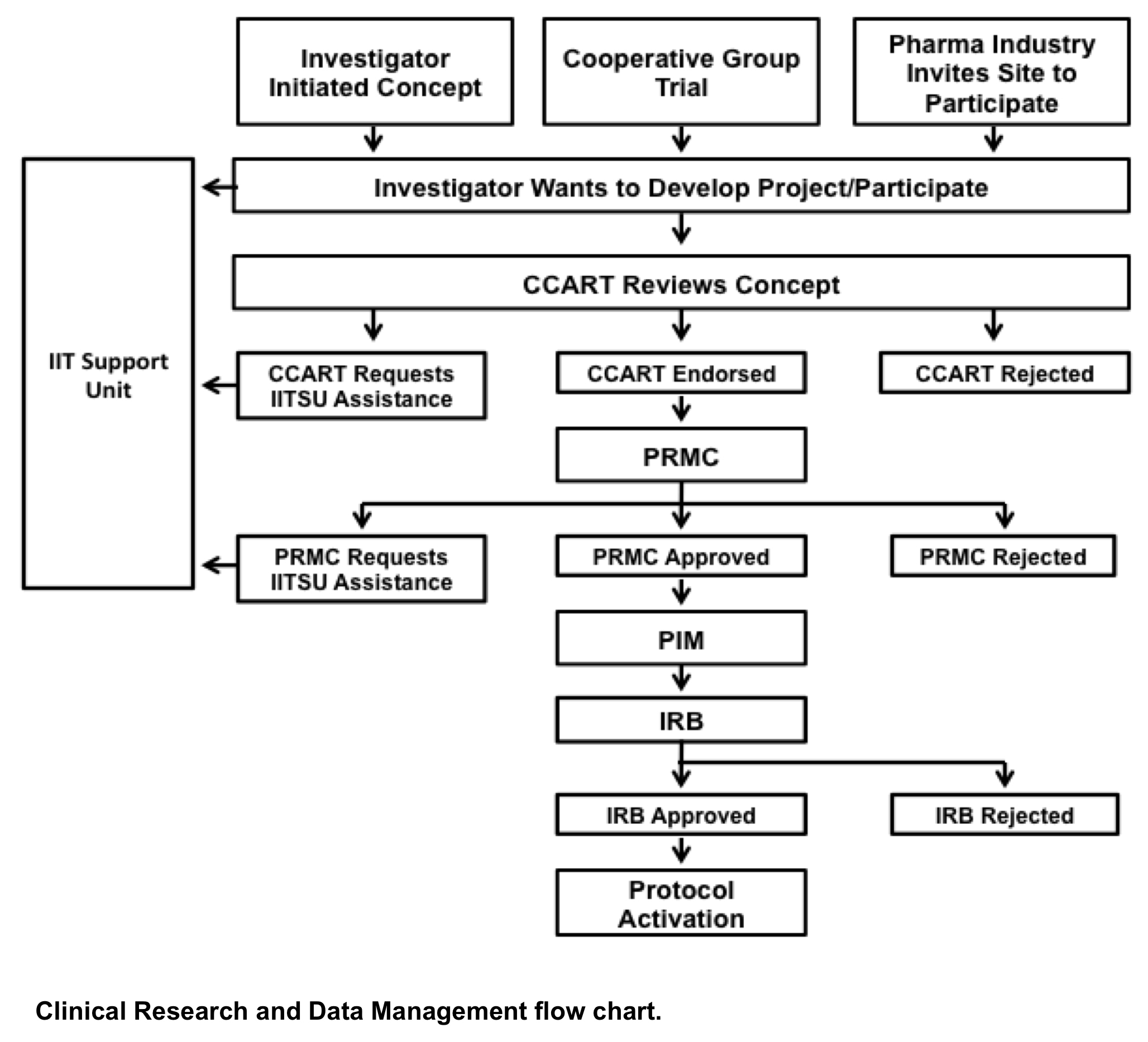
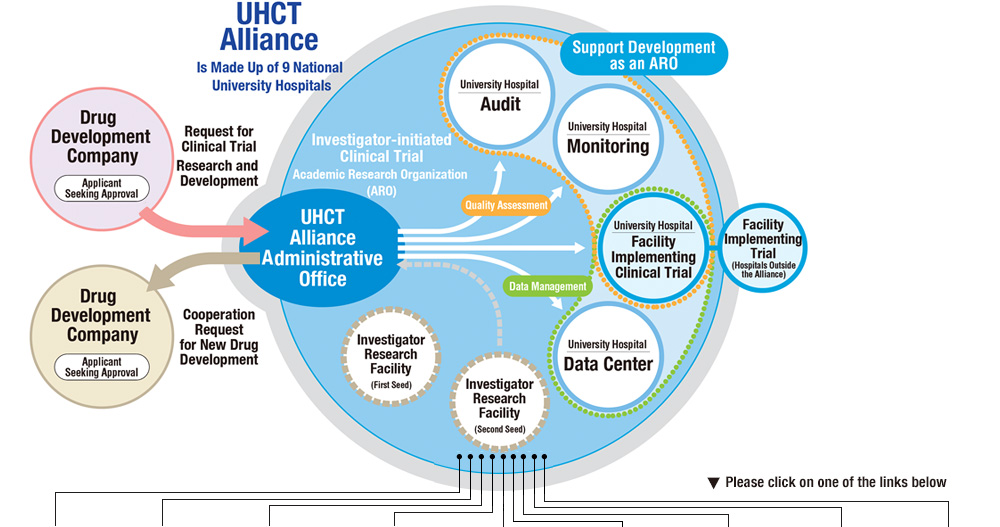
MJA on Twitter: "Value proposition of investigator‐initiated clinical trials conducted by networks … #freeaccess 1 week … "Investigator‐initiated trials run by clinical trial networks provide net economic benefits to health systems" https://t.co ...

Navigating the differences between Investigator- Initiated v. Sponsor-Initiated Clinical Trials – What are the special contract and administration issues. - ppt download

Improving and sustaining the site investigator community: Recommendations from the Clinical Trials Transformation Initiative - ScienceDirect
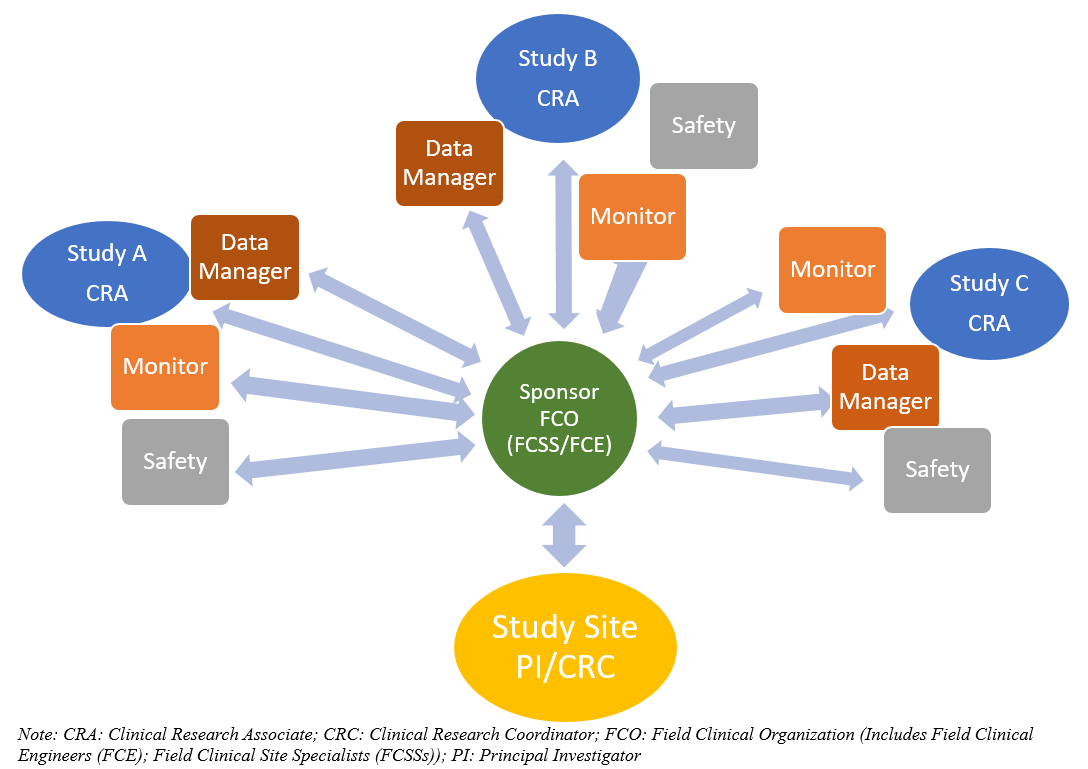
Sponsor-Site Communication in Device Trials: Evolution of a Dedicated Field Clinical Organization Throughout Study Execution - ACRP

Industry Viewpoints: Investigator-Initiated Studies – A Challenge for Supply with IMPs? - Clinical Trials Arena

Investigator-initiated trials of targeted oncology agents: why independent research is at risk? - Annals of Oncology
Investigator Initiated Trials (IIT) – Considerations and Guidance from the Perspective of Clinical Trial Supplies and GMP | ISPE | International Society for Pharmaceutical Engineering