Comparative analysis of two methods of data entry into electronic medical records: A randomized clinical trial (research letter) - Roman‐Belmonte - 2017 - Journal of Evaluation in Clinical Practice - Wiley Online Library

PDF) UK Medical Research Council and multicentre clinical trials: From a damning report to international recognition
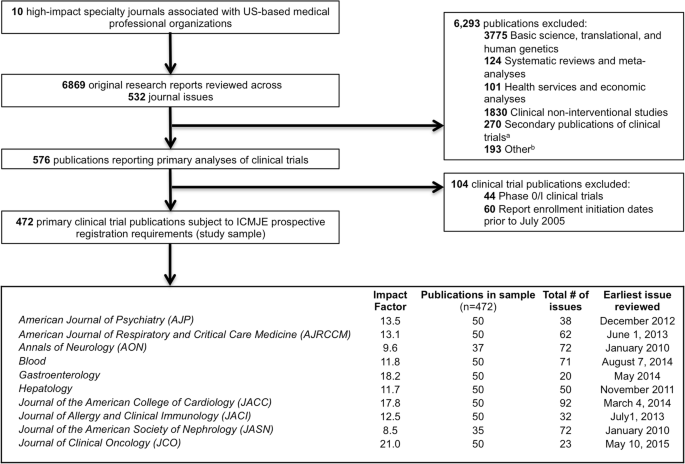
Adherence to the International Committee of Medical Journal Editors' (ICMJE) prospective registration policy and implications for outcome integrity: a cross-sectional analysis of trials published in high-impact specialty society journals | Trials